Modern academic research is largely a rather slow process taking small incremental steps. I’ve vented my frustration before about how dispiriting it can be to get lost in a fog of low-level research projects which often leave us more confused rather than enlightened. I thus feel I want to celebrate a rare occasion when I do feel a sense of completion of a substantial programme of research.
I was lucky enough to move to Belfast shortly after Kerr Graham and Aidan Cosgrove had completed their early work demonstrating the efficacy of Botulinum toxin injections first in hereditary spastic mice and then in children with cerebral palsy. Kerr had departed for Melbourne by the time I arrived but left Niall Eames, an orthopaedic surgeon, lined up to do some research to try and better understand the effect of the toxin. Given that the problem in CP is that the muscles are too short and that Botulinum toxin, by reducing the neural input to the muscle, allows them to elongate, we decided that we should do this by looking at the changes in muscle length. We thus started with some, by modern standards extremely crude, muscle length modelling of the gastrocnemius.

Response to botulinum toxin plotted against the pre-operative dynamic component (taken from Eames et al. 1999)
Having developed the model we applied it to a cohort of children with cerebral palsy having Botulinum toxin injections and were able to demonstrate that the action of the toxin was to reduce the “dynamic component” of reduced muscle length (see figure above). This makes a lot of sense as it is this component that is affected by the neural input to the muscle. The “fixed component” (contracture) is largely a consequence of changes to the composition and structure of the muscle and is unlikely to be affected by the toxin. The research also allowed us to understand that the variable response was largely due to children having a different dynamic component rather than of the toxin acting differently and led to reasonably simple prescription guidelines. Botulinum injections to the calf are most likely to be beneficial if the child has a large dynamic component (good range of passive dorsiflexion during physical examination but walking up on their toes). It further explained that the different response in children with diplegia and hemiplegia was also attributable to them having different magnitudes of dynamic component.
Armed with this understanding I was then able to work with the pharmaceutical company Ipsen to set up a cliniucal trail to establish the most appropriate dose of the toxin. We couldn’t find enough children to study in the UK so had to extend the study to five centres in Poland. We divided children into one of four groups and injected them with either a placebo or one of three different doses. We used the same modelling technique which we had developed for the earlier study to analyse the results and came to the conclusion that placebo didn’t work (very much) and that the middle dose was the most effective (see figure below). It was interesting that the biomechanical modelling came to clear logical conclusions whereas doctors’ subjective opinions were that the placebo was very nearly as effective as the drug and that they were so impressed by the “improvement” after placebo injection that they would have recommended repeating the process for two thirds of the children! (despite biomechanical evidence that the placebo had had no effect).
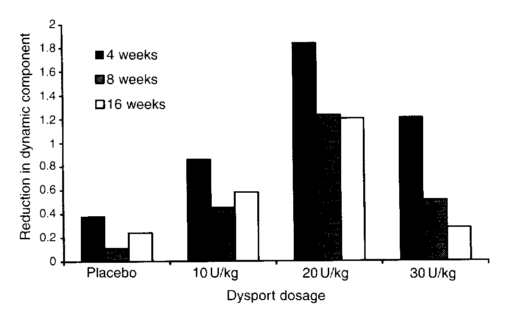
Reduction in dynamic component as a function of different doses of Botulinum toxin at 4, 8 and 16 weeks (Baker et al. 2002)
Having established the most appropriate dose on a single occasion the most obvious remaining question is, “How often should those injections be repeated?”. I’d moved to Melbourne to join Kerr by then and we applied to the Australian National Health and Research Council to fund a clinical trial to compare injections delivered either annually or every four months over a two year period. Reflecting on the biomechanics we recognised that the long term goal of the injections had more to do with preventing the development of secondary fixed contractures than on the immediate effect on the dynamic component. We would have to measure relatively small changes over a two year time span and thus devised a method to standardise the measurement of passive dorsiflexion range as much as possible.
Which brings me to the stimulus for writing this post in that the results of that study have just been published . The first conclusion is that passive range of dorsiflexion was maintained over the two year period by both injection regimes. We had no true control, because by this stage it wasn’t considered ethical to inject placebo over such a long period, but these measurements were taken over an age range in a child’s life during which preserving dorsiflexion range would be extremely unlikely without injections. The second conclusion was that the more regular injections where only slightly more effective in preserving dorsiflexion range and therefore that there doesn’t appear to be any particular benefit in injecting more regularly than once a year.
Thus after nearly twenty years of research based on the application of thoughtful biomechanics to a clinical problem we finally have clear evidence of which children to inject, how much toxin to inject and how often to repeat this. As one leader of the western world was once heard to comment under less auspicious circumstances, “Mission accomplished!”
Footnote
Trials like this take so long to organise that we were not actually the first group to complete a study to establish the most appropriate injection frequency. This was actually published about 5 years ago. It was a very similar study (it had been sponsored by Ipsen as a follow-on our to earlier work and I’d had some involvement in its planning before I left for Australia) and arrived at a very similar result. Rather than feeling that there was competition here though it highlights the scientific importance of repeating studies to confirm results. With such an emphasis on innovation in modern clinical research the need to repeat and confirm earlier results, which is an important part of the scientific process, can very often be overlooked.