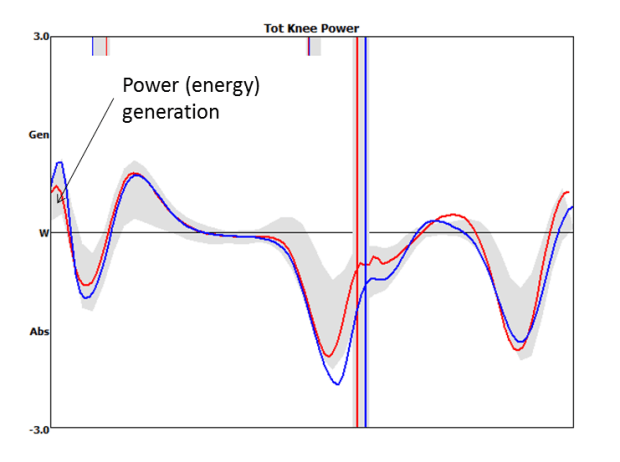
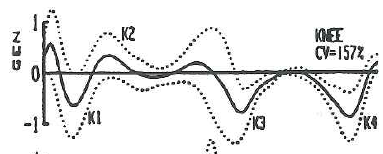
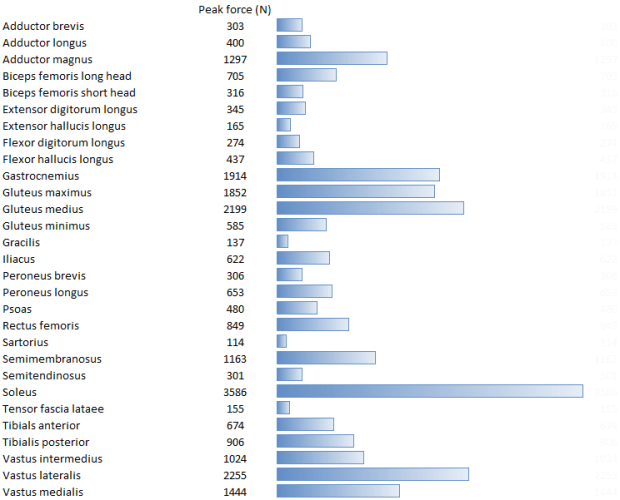
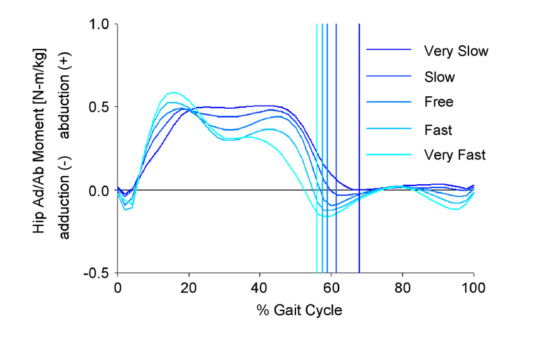
While I surfing the web doing a bit of background reading for last week’s post I came across this graph.
Ralston HJ (1958) Energy-speed relation and optimal speed during level walking. Int Z angew. Physiol. einschl. Arbeitsphysiol. 17 (8): 273-288.
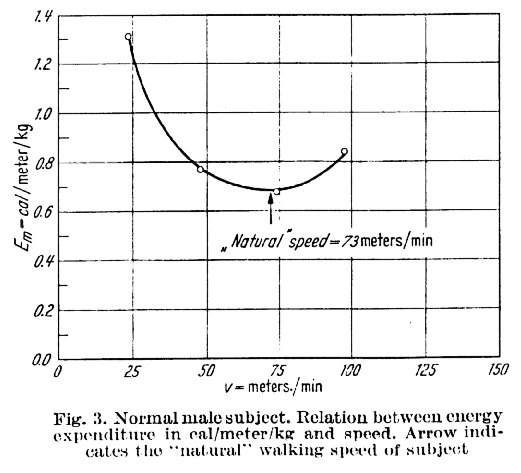
It’s another of the classic outputs of Verne Inman’s group, from Henry Ralston, and shows data for a healthy subject to support his hypothesis that we select our walking speed to minimise the energy cost of walking (the energy used to travel a certain distance). The hypothesis is so plausible that it has been almost universally accepted.
What interests me is that despite being so widely accepted I’ve never seen any suggestion of the mechanism through which we might achieve this. It’s a fairly basic principle of control theory that if we want to minimise any particular variable (such as distance walked for a given amount of energy) we need some way of measuring it. Thus it is very difficult to drive a car fuel efficiently if you just have a speedometer and a standard fuel gauge. If you add a readout to the dashboard telling you how much fuel you are using per kilometre travelled and the task becomes trivial. They should be compulsory in a fuel challenged world!
I’m not aware of any proprioceptive mechanism that would allow the brain to “know” how much energy it is using per unit distance walked. I can see that there are complex mechanisms regulating cardiac and pulmonary rate based primarily on carbon dioxide concentration in the blood which might allow us to sense how much energy we are using per unit time, but how can we possible sense how much energy we are using per unit distance. I’m not saying it’s impossible – the brain is a marvellous organ and it is possible that it integrates such a measure of energy rate (per unit time) with information about cadence and proprioception of joint angle and in order to derive a measure of energy cost (per unit distance). This is a complex mechanism however and certainly suggests that, as with so much in biology, whilst the basic hypothesis is extremely simple the mechanisms required to achieve this is far more complex than we might have imagined. As Ralston himself put it, “one of the most interesting problems in physiology is to elucidate the built in mechanism by which a person tends to adopt an optimum walking velocity such that energy expenditure per unit distance is a minimum”.
But this also makes me want to question the underlying hypothesis. Going back to the original paper (which you can read here), Ralston only produces data from one healthy subject and one amputee to support his hypothesis. I’m not aware of many others having explored the hypothesis on an individual level (the conclusion that the self-selected walking speed is close to speed of minimum energy cost for a sample does not mean that the relationship holds for individuals within that sample). I’d be interested to hear from readers of papers that have investigated this relationship in more detail.
The other point that Ralston made which is almost always overlooked is that the curve is “almost flat”. The curve only looks so steep because it has been plotted over such a wide range of values (from 0 through to 150m/s). Just looking at the data plotted I’d suggest that the speed can range from about 56 to 84 m/min whilst the energy cost remains within 5% of the minimum energy cost value. This is almost certainly within the range of measurement error for a variable such as energy cost. In other words the really remarkable thing about the energy curve is that it allows us to walk over quite a range of speeds without having a measureable effect on our energy cost. It is interesting that Ralston managed to make this point and suggest that we select walking speed to minimise energy cost in the same paper!